Atessia supports its clients daily in the practical modalities of implementing the French early and compassionate access system, whose subtleties require some explanations.
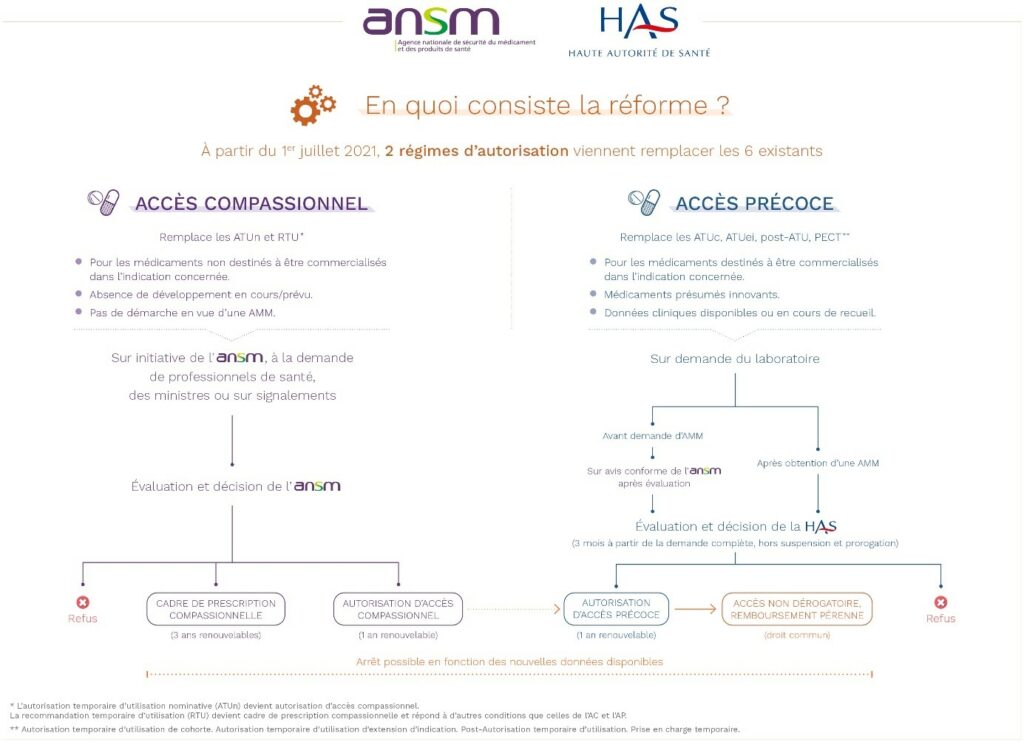
On July 1, 2021, the new early and compassionate access system was introduced through 2 decrees, supplemented by 4 orders, with immediate effect. This new system is based on 2 mechanisms for access and coverage by health insurance:
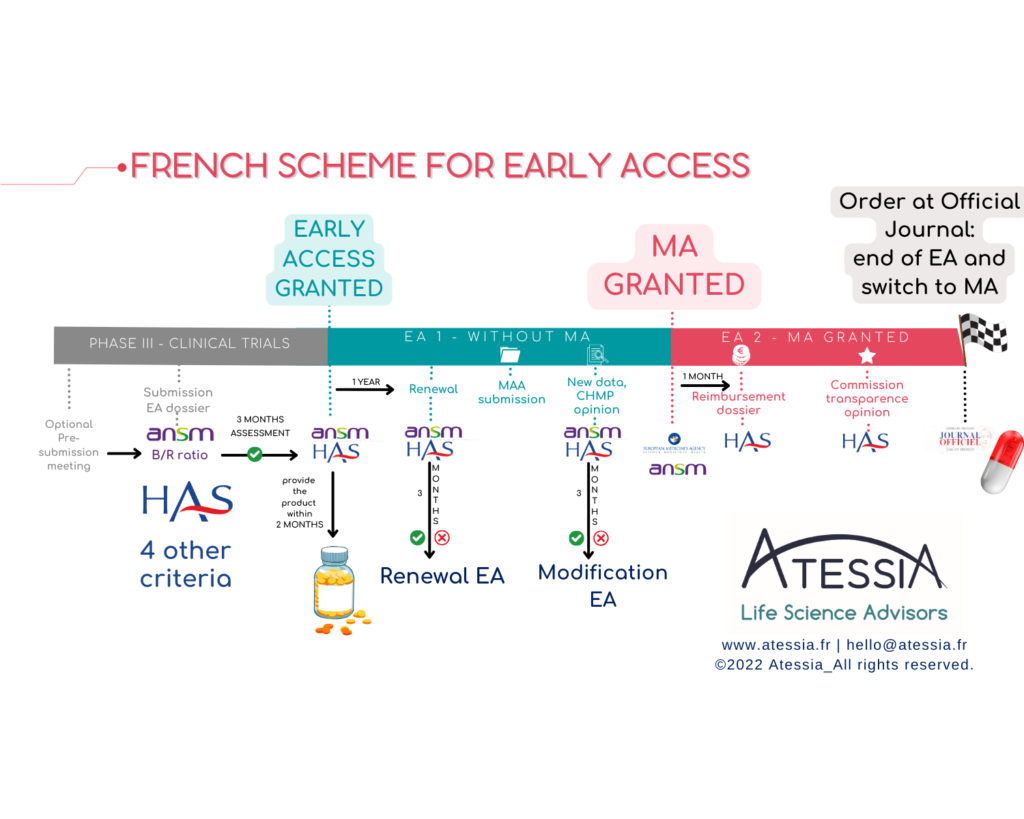
- Early Access (AAP)
Firstly, early access, which targets medicinal products that meet an unmet therapeutic need and may be innovative. The laboratory submits a request for early access authorization (AAP) to the High Authority for Health (HAS) and, for medicinal products not yet authorized under a Marketing Authorization (AMM), to the National Agency for the Safety of Medicines and Health Products (ANSM).
These authorizations can apply to:
- A medicinal product prior to obtaining the AMM in the considered indication (Pre-AMM AAP = AP1),
- A medicinal product that already has an AMM in the considered indication, prior to common law coverage by health insurance (Post-AMM AAP = AP2) Interestingly, the product may or may not have an AMM for another indication. As per HAS doctrine, granting early access authorization is reserved for certain specialties meeting the following 5 cumulative eligibility criteria:
1. Strongly presumed efficacy and safety in the considered indication.
2. The disease to be treated is severe, rare, or disabling.
3. There is no “appropriate treatment.”
4. The implementation of treatment cannot be delayed.
5. The medicinal product is presumed to be innovative.
The authorities examine each of these criteria separately, in a relatively strict manner.
This system also requires concrete commitments from laboratories, which should not be underestimated and need to be weighed with the parent company.
- From a REGULATORY standpoint: the laboratory must commit to filing an AMM request within 2 years for an AAP1 or a request for registration within the month following the AMM’s approval for an AAP2. Thus, the timing of the filing is crucial in the project.
- From a LOGISTICAL standpoint: the laboratory makes the product available within 2 months following the granting of the AAP (PUI) and ensures it can supply the product to allow continuity of treatments initiated during the entire AAP, for a minimum period of one year (including 3 months of coverage).
- From a FINANCIAL standpoint: the laboratory implements a PUT-RD, for data collection and transmission of periodic summary reports. The laboratory finances this data collection (cf. agreement to be signed with health establishments).
- The pharmaceutical laboratory is also required to assist prescribers in entering and monitoring the collection of real-life follow-up data of the medicinal product, providing them with the necessary means.
Two types of compassionate access:
This system targets two distinct cases, both involving a medicinal product to treat patients with diseases without appropriate treatment in a given therapeutic indication, without being intended to obtain an AMM in France. The requests are managed only by the National Agency for the Safety of Medicines and Health Products (ANSM).
- Either this compassionate access is requested for an unauthorized and unavailable medicinal product in France by a hospital prescriber for a specifically named patient, provided that the ANSM can presume a favorable benefit/risk ratio for a severe, rare, or disabling disease: this is an individual and nominative compassionate access authorization (AAC).
- Or it involves the regulation of a practice, initiated by the ANSM, to secure an off-label prescription practice of a medicinal product available in France, with an AMM for other indications, when it is subject to a well-established off-label prescription on French territory: this is a compassionate prescription framework (CPC).
Exemptions to compassionate access have been foreseen in the following cases:
- Allowing nominative access to medicinal products in development for the indication: this is a “very early” compassionate access.
The grant by the ANSM is subject to several eligibility conditions, which brings this system closer to early access and can be the gateway to it:
- The implementation of the treatment cannot be delayed;
- The patient cannot participate in any ongoing research;
- The company marketing the medicinal product must commit to filing an early access request within 12 months following the first “pre-precoce compassionate” authorization (18 months for rare diseases).
For these mechanisms, the designation of a laboratory operating a medicinal product may be necessary, to ensure, if necessary, the import/distribution, pharmacovigilance, quality complaints, or medical information.
The laboratories now have several years of experience with these new systems, and the emerging trends show the authorities’ willingness to make innovative medicinal products available to French patients and to respond to the personal situations of patients in therapeutic dead ends.
Article written by Caroline LECUELLE, Consultant in Regulatory Affairs & Pharmaceuticals